Introduction
Medication non-adherence, i.e., patients not taking medications as prescribed, diminishes clinical outcomes and quality of life. This phenomenon is endemic, leading to an estimated 200,000 premature deaths and 125 billion Euros of excess healthcare costs each year in Europe alone [1].
Medication adherence (MA) consists of three interrelated phases: initiation (first dose taken), implementation (daily regimen management) and discontinuation (last dose taken) [2]. In Switzerland, half of the population aged 15 years and older takes at least one medication per week [3]. Worldwide, studies showed that most patients (69–96%) initiate their prescribed treatments correctly, but 30–50% do not implement them optimally. Within 2 years of their first prescription, more than 50% of patients discontinue use of their prescribed medication [1].
The high prevalence and consequences of medication non-adherence have attracted considerable attention over the past 30 years. Researchers, clinicians and policymakers search for improved methods to assess this phenomenon and its determinants, along with effective MA-enhancing multifaceted interventions tailored to patients’ needs [4].
In 2011, the UK’s National Health Service (NHS) introduced the largest-ever national evidence-based MA intervention: the New Medicine Service (NMS) [5]. After certain new medications are prescribed, NHS pharmacists identify and assess any adherence problems and their causes. While increasing quality-adjusted life-years per patient, the NMS has led to reduced costs compared with standard care [6].
While evidence on addressing medication non-adherence is available, translation of that evidence into real-world clinical practice remains challenging. In Switzerland, no adherence-focused national programs or guidelines have been developed, implemented or evaluated [7].
Therefore, the launch of the European Network to Advance Best practices and technoLogy on medication adherencE (ENABLE), a Cooperation in Science and Technology (COST) Action (https://www.cost.eu/actions/CA19132/) in 2020 was well-timed [8]. Linking stakeholders across 39 European countries, including Switzerland, ENABLE aims to drive the implementation of MA-enhancing technologies into clinical practice.
Launch of the Swiss COST ENABLE Network
On March 18th, 2022 Switzerland’s ENABLE country group launched a pioneering initiative to set priorities for implementing MA interventions in real-world settings. An online conference was held to: 1) connect Swiss and international stakeholders; and 2) initiate MA priority-setting, particularly regarding implementation of MA interventions and technologies.
Seventy-five participants including researchers, clinicians, healthcare industry delegates and one policymaker, from 34 countries took part. Keynote lectures and an interprofessional roundtable highlighted the state-of-the-art MA understanding regarding the place for digital technology in real-world settings, alongside strategies to address non-adherence. Priorities to move toward a more favourable, multilevel ecosystem for developing, implementing and maintaining MA interventions in Switzerland were discussed. While the highest priority was identified as support to the implementation of MA interventions in real-world settings, education, research and policy measures were also highlighted (Supplementary File S1).
The ideas discussed were organized into a framework (Figure 1)—partly for a systematic elaboration of strategies to address MA at a national level, partly to move towards an integrated, patient-as-partner healthcare system.
FIGURE 1
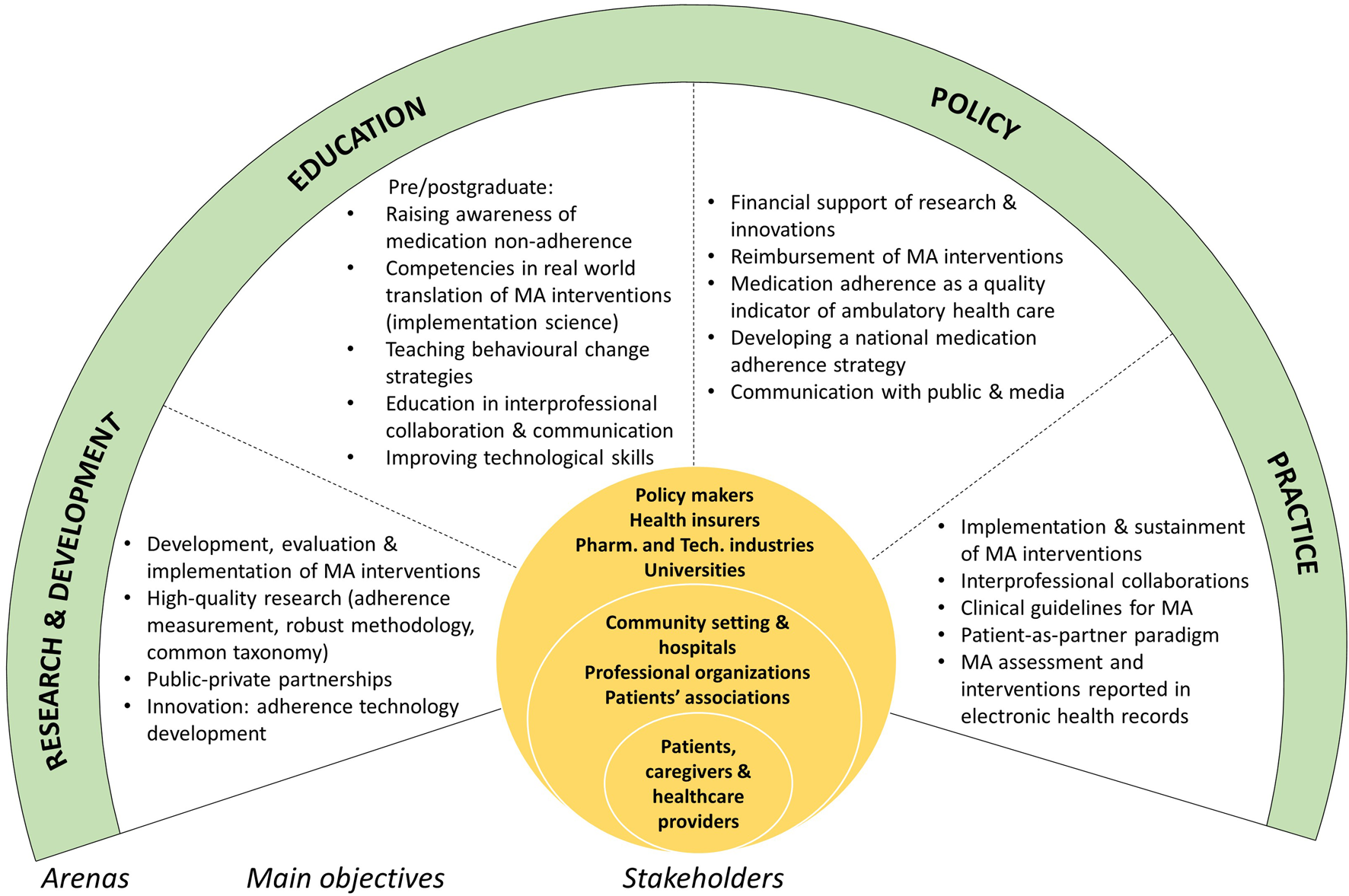
Framework for addressing medication adherence in a complex, multilevel ecosystem: arenas, main objectives and stakeholders. European Network to Advance Best practices and technoLogy on medication adherencE (ENABLE), a Cooperation in Science and Technology (COST) Action, Switzerland, 2022.
Priority Setting to Implement MA Interventions in Switzerland
While MA is a major public health concern that warrants high-priority treatment by health authorities, it remains absent from Swiss policymakers’ agendas. As a first step, raising awareness for MA and promoting research are needed to change the mindset and attitudes of the population, health care professionals (HCPs), and all other stakeholders.
Prerequisites for MA Intervention Implementation
Multilevel Ecosystem
To achieve synergies, stakeholders must work interprofessionally, fine-tuning their actions, and engaging their responsibilities to collaboratively monitor and maximize MA in the ecosystem. Connecting innovative ecosystems in Switzerland (e.g., funding agencies, start-up incubators) and public-private-partnerships will foster further MA development (e.g., new MA technologies).
While reimbursement of MA interventions -by health insurers- will enhance adoption of these interventions in clinical practice, the scientific basis to support reimbursement should go beyond demonstration of MA improvement. A clear value statement is needed in which the effects of MA interventions guarantee favorable clinical outcomes [9].
Patient-As-Partner
Along with shared decision-making and therapeutic education, the patient-as-partner paradigm is critical to raise MA awareness and for mutual learning.
However, efforts should not only target the individual level. Building trusting partnerships and communication across all HCPs and patients reinforces MA by decreasing conflicting information and ensuring continuity of care. Emphasizing MA interventions, behavioural training and interprofessional competencies should be integrated within the training curricula of all involved HCPs (i.e., including physicians, nurses, psychologists, pharmacists) to develop competencies vital to MA management in clinical practice. Integrated care models powered by digitalization provide fertile ground for these efforts and innovations.
Monitoring Medication Adherence
Measuring MA and its determinants in routine practice enables researchers to know when patients have MA difficulties or underlying non-adherence risk factors and provides an informed basis for clinical decision-making. In routine practice, adherence should be treated both as a patient’s fifth vital sign and as a system-level indicator of quality of care. Technology (e.g., smartphone apps, electronic adherence monitors) can be helpful, but must be designed to match patients’ and HCPs’ preferences.
Investing in Translation From Research to Real-World Settings
Internationally, research evidence underscores the value of multilevel interventions, monitoring and equipping HCPs to address MA [1, 4]. However, in Switzerland, implementation of MA interventions in clinical practice is limited. Assisting the uptake of research evidence into clinical practice will require a systematic implementation science approach [10].
Next Steps to Support the Translation of MA Interventions into Swiss Clinical Practice
First, key interprofessional cross-sectoral stakeholders need to be identified to address MA in the multilevel ecosystem and to determine contextually appropriate multilevel MA interventions. Every step will need to fit within a national Swiss MA strategy.
Second, efforts towards coordinated care models must be strengthened and tailored to facilitate MA management. Technology-based innovations and digital health infrastructure (e.g., electronic medical records, teleconsultation) and use of new technologies (e.g., digital pill boxes, smart inhalers) and methodologies (e.g., artificial intelligence) have the potential to facilitate or contribute directly to interventions. Importantly, technology literacy needs to be concurrently enhanced at the patient and HCP levels.
Third, contextually appropriate implementation strategies (e.g., development of clinical guidelines, patient/consumer involvement) need to be integrated using a multilevel approach.
Fourth, research funding schemes must be adapted to provide opportunities for MA research (e.g., MA is prioritized by the Innovative Medicines Initiative, an EU public-private-partnership). Researchers should be encouraged to expand their foci from traditional efficacy/effectiveness research to include evaluation of implementation strategies for MA interventions in real-world settings.
Finally, to maximize impact, the synergies between bottom-up and top-down approaches, public/private initiatives and interprofessional collaborations are required to power much-needed change.
Conclusion: A Framework for Change
The Swiss COST ENABLE conference built on its participants’ critical knowledge, competencies and experience of interprofessional dynamics to define priorities to tackle MA as a major public health issue. One point was clear: translating MA-relevant evidence into real-world outcomes demands multilevel approaches.
Through the Swiss ENABLE conference, diverse presenters and participants committed to raising MA awareness. Regarding the knowledge and practice gap, an initial consensus was reached on three early steps: provide the foundations for MA-supportive ecosystems, translate effective interventions into clinical practice and promote research into MA-enhancing technologies. The Swiss strategies discussed here will potentially inform research activities, policies and implementation of MA interventions across Europe.
Statements
Author contributions
SDG and MS conceptualized and organized the conference. After the conference, all authors discussed the main themes to be included in the manuscript. CB and JR drafted the paper and SDG, MS, and AD reviewed it. All authors gave feedback on the draft. All authors agreed upon the final version of the paper.
Funding
The event was supported by the Swiss National Science Foundation (IZSEZ0_208877) and the ENABLE COST Action (19132).
Acknowledgments
We sincerely thank ENABLE and Swiss National Science Foundation for funding, Juliane Barnick, Miroslawa Rebetez and AVL Interpreters for logistical support and all participants from Switzerland and abroad. In addition, we thank Chris Shultis for his English editing.
Conflict of interest
Author MO was employed by the company Novartis Pharma AG.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Author disclaimer
The statements in this commentary represent the personal view of DA, it does not engage the position and commitment of the Federal Department of Home Affairs, Federal Office of Public Health Division, Switzerland.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.ssph-journal.org/articles/10.3389/ijph.2022.1605204/full#supplementary-material
References
1.
KhanRSocha-DietrichK. Investing in Medication Adherence Improves Health Outcomes and Health System Efficiency: Adherence to Medicines for Diabetes, Hypertension, and Hyperlipidaemia. Paris: OECD Publishing (2018).
2.
VrijensBDe GeestSHughesDAPrzemyslawKDemonceauJRupparTet alA New Taxonomy for Describing and Defining Adherence to Medications. Br J Clin Pharmacol (2012) 73(5):691–705. 10.1111/j.1365-2125.2012.04167.x
3.
Office-Federal-de-la-Statistique. Enquête suisse sur la santé 2017: Une personne sur deux en Suisse prend des médicaments chaque semaine (2019). Available at: https://www.seco.admin.ch/seco/fr/home/seco/nsb-news.msg-id-74151.html (Accessed March 25, 2022).
4.
NieuwlaatRWilczynskiNNavarroTHobsonNJefferyRKeepanasserilAet alInterventions for Enhancing Medication Adherence. Cochrane Database Syst Rev (2014) 2014(11):CD000011. 10.1002/14651858.CD000011.pub4
5.
National Health Service. Advanced Service Specification - New Medicine Service (2021). Available at: https://www.england.nhs.uk/publication/advanced-service-specification-nhs-new-medicine-service-nms/ (Accessed April 14, 2022).
6.
ElliottRATanajewskiLGkountourasGAveryAJBarberNMehtaRet alCost Effectiveness of Support for People Starting a New Medication for a Long-Term Condition through Community Pharmacies: an Economic Evaluation of the New Medicine Service (NmS) Compared with normal Practice. Pharmacoeconomics (2017) 35(12):1237–55. 10.1007/s40273-017-0554-9
7.
KostalovaBRibautJDobbelsFGerullSMala-LadovaKZulligLLet alMedication Adherence Interventions in Transplantation Lack Information on How to Implement Findings from Randomized Controlled Trials in Real-World Settings: A Systematic Review. Transpl Rev (2021) 36:100671. 10.1016/j.trre.2021.100671
8.
Van BovenJFTsiligianniIPotocnjakIMihajlovicJDimaALNabergoj MakovecUet alEuropean Network to advance Best Practices and Technology on Medication Adherence: mission Statement. Front Pharmacol (2021) 12:748702. 10.3389/fphar.2021.748702
9.
BartholomewTNaciHRobertsonESchmidtH. Use of Adherence Monitoring in Drug Contracts Tied to Outcomes: Put Patients First. BMJ (2022) 376:e062188. 10.1136/bmj-2020-062188
10.
PetersDHAdamTAlongeOAgyepongIATranN. Republished Research: Implementation Research: what it Is and How to Do it: Implementation Research Is a Growing but Not Well Understood Field of Health Research that Can Contribute to More Effective Public Health and Clinical Policies and Programmes. This Article Provides a Broad Definition of Implementation Research and Outlines Key Principles for How to Do it.Br J Sports Med (2014) 48(8):731–6. 10.1136/bmj.f6753
Summary
Keywords
medication adherence, multi-level interventions, priority setting, COST action ENABLE, Swiss research, implementation science, interprofessionality, healthcare policy
Citation
Bandiera C, Ribaut J, Dima AL, Allemann SS, Molesworth K, Kalumiya K, Käser F, Olson MS, Burnier M, van Boven JFM, Szucs T, Albrecht D, Wilson I, De Geest S and Schneider MP (2022) Swiss Priority Setting on Implementing Medication Adherence Interventions as Part of the European ENABLE COST Action. Int J Public Health 67:1605204. doi: 10.3389/ijph.2022.1605204
Received
09 July 2022
Accepted
18 July 2022
Published
12 August 2022
Volume
67 - 2022
Edited by
Nino Kuenzli, Swiss Tropical and Public Health Institute (Swiss TPH), Switzerland
Updates
Copyright
© 2022 Bandiera, Ribaut, Dima, Allemann, Molesworth, Kalumiya, Käser, Olson, Burnier, van Boven, Szucs, Albrecht, Wilson, De Geest and Schneider.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sabina De Geest, sabina.degeest@unibas.ch; Marie P. Schneider, marie.schneider@unige.ch
†These authors share first authorship
‡These authors share last authorship
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.